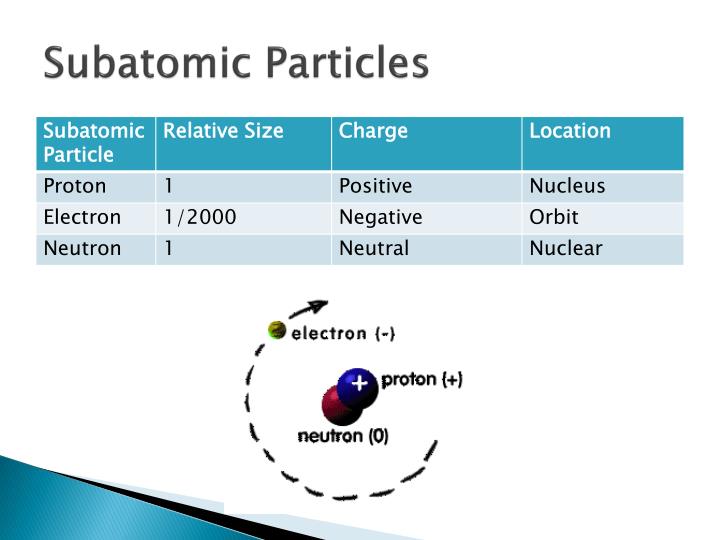

The electron has a charge, e, of 1.6 ×10 -19 Coulombs (a unit of electrical charge named for French physicist, Charles-Augustin de Coulomb ). The charges of all particles are traditionally measured in terms of the size of the charge of the electron. Thomson gave it the name corpuscle, which was later changed to electron. The charge-to-mass ratio was found to be relatively large, and independent of the gas used in his experiments, which indicated to him that he had found a true particle. Thomson (1856 –1940), in 1897, measured the velocity and charge-to-mass ratio of these particles. Other scientists had deduced the existence of a negatively charged particle in what were called cathode rays (and which are now known to be beams of electrons). The first subatomic particle to be discovered was the electron. All forces, including gravity, are thought to be mediated by particle exchanges. That is, when two electrons collide, they do not simply bounce off of each other like two billiard balls: they exchange a photon (one of the mediator particles). These mediator particles enable the matter particles to interact with each other. The other elementary particles are mediators of the fundamental forces. The most famous baryons are protons and neutrons. Baryons and mesons are combinations of quarks and are considered subatomic particles. Examples of these particles include quarks (which make up protons and neutrons) and electrons. There are two types of elementary particles.

However, the list of subatomic particles has now been expanded to include a large number of elementary particles and the particles they can be combined to make. Early in the twentieth century, electrons, protons, and neutrons were thought to be the only subatomic particles these were also thought to be elementary (i.e., incapable of being broken down into yet smaller particles). Subatomic particles are particles that are smaller than an atom.
